FULL UNDERSTANDING OF THE STAINLESS STEEL PROCESSING
Unlike all other areas, only the construction of a pharmaceutical “thermocompressor” for the production of Water for Injection claims the most extreme care. It’s due to the “purity” required throughout the transformation process, from raw water to WFI. The BRAM-COR observance of the GMP reference standards supports the quality of the STMC distiller manufacturing.

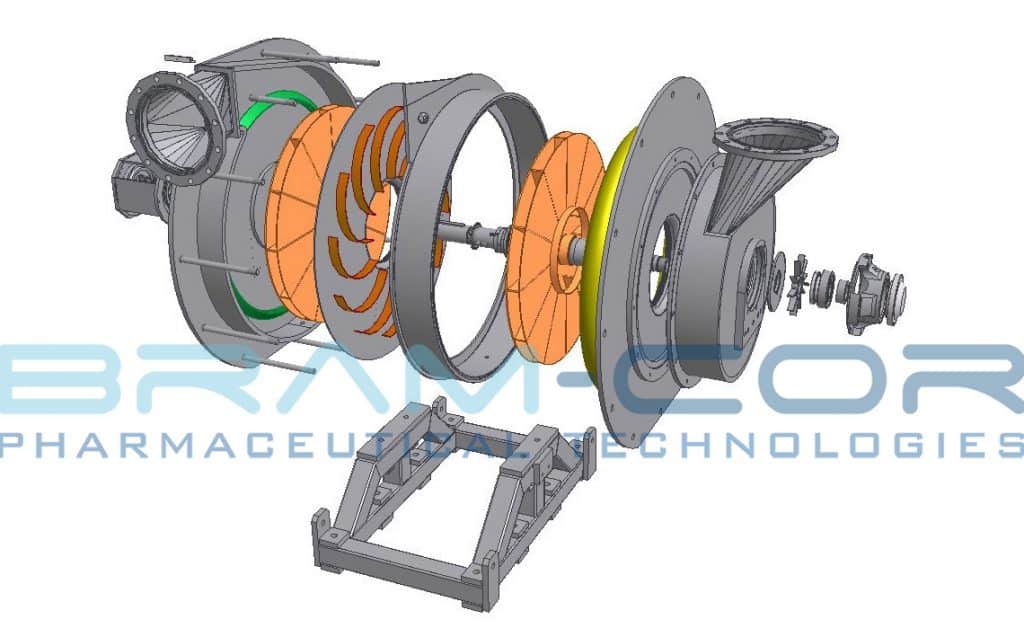
The experience and passion for stainless steel processing enable BRAM-COR to care for quality in vapor compression distiller manufacturing. Therefore, the construction of each STMC distiller follows cGMP procedures. In Parma, the location of BRAM-COR headquarters, qualified welders perform regularly BRAM-COR Sanitary Piping procedures (we use AISI 316L Stainless steel for piping and AISI 304 for the frameworks only).


Non-destructive tests are performed during construction. A single tag identifies each component, engraved on metal plate, for the total traceability in the relevant technical documentation.

Functional testing is ensured by automation experts, with special care for monitoring of critical parameters. Our project management includes documentation, inspection and field testing.
SOME DISTINCTION IN BRAM-COR VAPOR COMPRESSION MANUFACTURING
In the States the Pharmaceutical and Biotech Company are mostly installing the Vapor Compression technology by USA companies, which have, depending from the suppliers, some disadvantages:
– Fast rotation. The rotation speed of compressor can reach 11.000 RPM, against the 3800 RPM of the BRAM-COR compressor, with stress problems in the shaft and in the compressor mechanisms (subjected to breakages) and lack of control on the evaporation speed, fundamental to grant the proper separation between dry pure steam and wet particles with contaminants.
– Horizontal configuration. Some other VCs are not born to be “pharmaceutical”, as the horizontal ones (knotty, not natural and not green), that were born for desalinization in the sea-boats. Their structure is in fact the forcing of the circulation, so they require many avoidable devices as sprays, some re-circulations pumps, bundles etc.. (subjected to breakages, as all mechanisms). Consequently this type of VC needs more frequent interventions/maintenance, spare parts changes, and very pure feed water (in contrast with the benefit that VC technology allows against ME stills).
– Direct coupling. The direct coupling, with lubricating oil circuit (dirty), at the same level of the circuit of the PS (clean-pharmaceutical), is offered as a benefit, but it is causing risk of cross contamination (especially but not only, for the VCs designed in the archaic vacuum configuration), as the dirty and the clean circuit are at the same level, with the possibility that some oil can reach the clean steam area. In these machines is in fact also present a “conduit”, closed to the impeller, to discharge the oil that can arrive to the WFI circuit. The direct coupling is also dangerous because the shaft is subjected to strong solicitations and stress, being held in just one side. See also “Advantage of the belt driven compressor in vapor compression still”.


Click here to see other Bram-Cor pharmaceutical lines (water treatment, processing, filling and packaging systems).
